Epithelial to Mesenchymal Transition
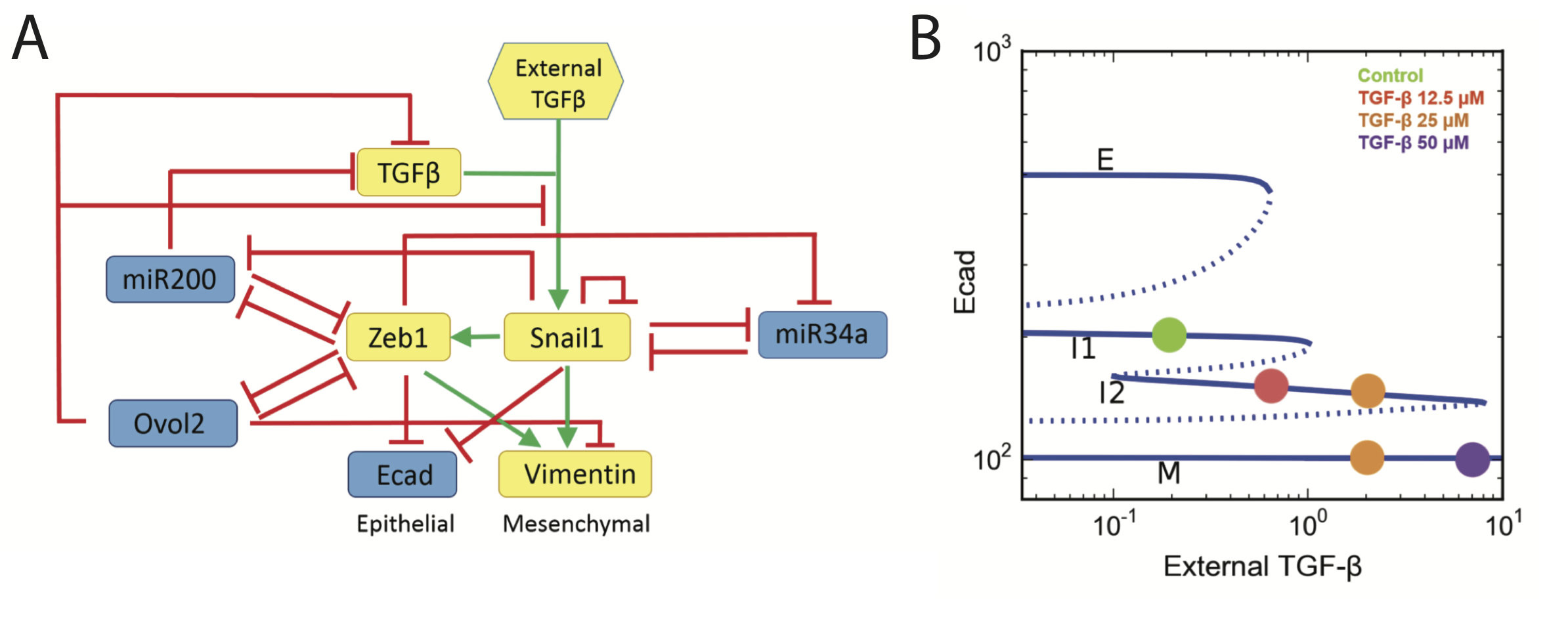
Epithelial to mesenchymal transition (EMT) describes the process by which cells lose markers and functions indicative of an epithelial state and gain those of a mesenchymal state. This process plays a crucial role during normal development and cancer progression. Our work on EMT focuses on modeling the cell fate transitions and studying the complex transcriptional dynamics that govern these transitions. We have predicted the existence of a new intermediate state between epithelial and mesenchymal fates via mathematical and dynamical systems analysis and verified such an intermediate state experimentally by flow cytometry. Intermediate states can increase the robustness of the system as well as its dynamical range. We have been able to predict how a delicate balance between EMT and the reverse transition (MET) is maintained in vivo.
At the transcriptional level, we can assess the importance of particular EMT-related genes using in silico tools, and in doing so improve our understanding of the topology and dynamics of the EMT network. Studying particular transcriptional motifs (such as a proposed mutual inhibition loop between Ovol2 and Zeb1) within the larger gene regulatory network and in a multiscale framework (i.e. coupled to cell- and tissue-level effects) enables a more complete map to be drawn of cell fate control via reversible EMT.
Our current and ongoing research in this area focuses on identifying the regulators of EMT and modeling their dynamics. In close collaboration with experimental biologists, we continue to dissect cellular heterogeneity and cell fate plasticity through experiment and theory.